Enterprise Data Warehouse, Built for Life Sciences
DATAstream™
A centralized data foundation for enterprise reporting, analytics, and insight.
DATAstream™:
Life sciences organizations generate large volumes of data across CROs, EDCs, labs, commercial platforms, and internal systems. That data is often fragmented across vendors and programs, making it difficult to access, reconcile, and reuse.
DATAstream™ addresses this challenge by bringing enterprise data together into a single, governed foundation. It enables organizations to reduce fragmentation, improve visibility, and support analytics and decision-making across clinical, commercial, and operational teams.
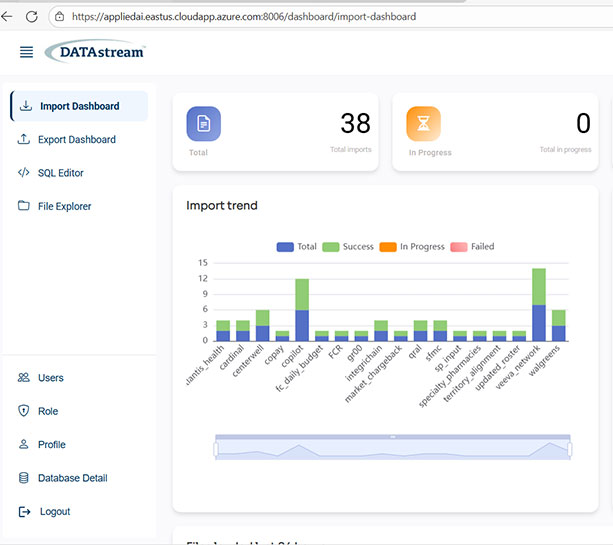
Built for Regulated Enterprise Data
The Impact of DATAstream™
Core DATAstream™ Capabilities
Build a Data Foundation That Scales
Whether consolidating enterprise reporting, enabling advanced analytics, or reducing data fragmentation, DATAstream™ provides the structure and scalability required for modern life sciences organizations.